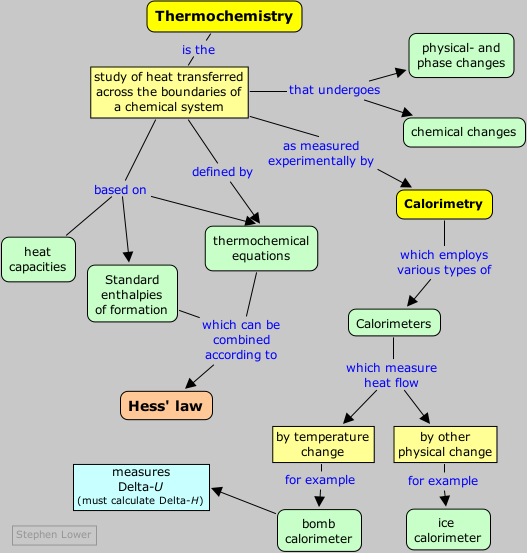
Thermochemistry - the study of energy changes that occur during chemical reactions and changes in state
Chemical potential energy - the energy stored in the chemical bonds of a substance
Heat (q) - the energy that transfers from one object to another because of a temperature difference between them. (heat always flows from a warmer object to a cooler object)
System - the part of the universe on which you focus your attention
Surroundings - everything else in the universe but the system
Law of conservation of energy - in any chemical or physical process, energy is neither created nor destroyed
Endothermic process - absorbs heat from surroundings.( In an endothermic process, the system gains heat as the surroundings cool down).
Exothermic process - releases heat to surroundings. (In an exothermic process the system loses heat as the surroundings heat up).
Calorie - the quantity of heat needed to raise the temperature of 1g pure water 1 degree C. 1 calorie = 1 kilocalorie= 1000 calories = 4.184 Joules
Heat capacity - the amount of heat needed to increase the temperature of an object exactly 1 degree C. (It depends on both the mass and the chemical composition of an object)
Specific heat - the amount of heat it takes to raise the temperature of 1g of a substance 1 degree C
Calorimetry - the precise measurement of the heat flow into or out of a system for chemical and physical processes.( In calorimetry, the heat released by a system is equal to the heat absorbed by the surroundings. Conversely, the heat absorbed by a system is equal to the heat released by its surroundings).
Enthalpy (H) - the heat content of a system at constant pressure
Thermochemical equation - a chemical equation that includes the enthalpy change (in a chemical equation, the enthalpy change for the reaction can be written as either a reactant or a product)
Heat of reaction - the enthalpy change for the chemical equation exactly as it is written
Heat of combustion - the heat of reaction for the complete burning of one mole of a substance
Molar heat of fusion - the heat absorbed by one mole of a solid substance as it melts to a liquid at a constant temperature
Molar heat of solidification - the heat lost when one mole of a liquid solidifies at a constant temperature
Molar heat of vaporization - the amount of heat necessary to vaporize one mole of a given liquid
Molar heat of condensation - the amount of heat released when one mole of vapor condenses at the normal boiling point (the quantity of heat absorbed by a vaporizing liquid is exactly the same as the quantity of heat released when the vapor condenses)
Molar heat of solution - the enthalpy change caused by dissolution of one mole of a substance (During the formation of a solution, heat is either released or absorbed)
Calorimeter - an insulated device used to measure the absorption or release of heat in chemical or physical processes
Hess's law of heat summation - if you add two or more thermochemical equations to give a final equation, then you also can add the heats of reaction to give the final heat of reaction (Hess's law allows you to determine the heat of reaction indirectly)
Standard heat of formation - the change in enthalpy that accompanies the formation of one mole of a compound from its elements with all substances in their standard states at 25 degrees celsius (For a reaction that occurs at standard conditions, you can calculate the heat of the reaction by using standard heat of formation)
Change in enthalpy - The heat gained or lost by a system at constant pressure. in this textbook, heat and enthalpy change are the same
Joules - unit for energy. One joule of heat raises the temperature of 1 g of pure water 0.2390 degree C
Chemical potential energy - the energy stored in the chemical bonds of a substance
Heat (q) - the energy that transfers from one object to another because of a temperature difference between them. (heat always flows from a warmer object to a cooler object)
System - the part of the universe on which you focus your attention
Surroundings - everything else in the universe but the system
Law of conservation of energy - in any chemical or physical process, energy is neither created nor destroyed
Endothermic process - absorbs heat from surroundings.( In an endothermic process, the system gains heat as the surroundings cool down).
Exothermic process - releases heat to surroundings. (In an exothermic process the system loses heat as the surroundings heat up).
Calorie - the quantity of heat needed to raise the temperature of 1g pure water 1 degree C. 1 calorie = 1 kilocalorie= 1000 calories = 4.184 Joules
Heat capacity - the amount of heat needed to increase the temperature of an object exactly 1 degree C. (It depends on both the mass and the chemical composition of an object)
Specific heat - the amount of heat it takes to raise the temperature of 1g of a substance 1 degree C
Calorimetry - the precise measurement of the heat flow into or out of a system for chemical and physical processes.( In calorimetry, the heat released by a system is equal to the heat absorbed by the surroundings. Conversely, the heat absorbed by a system is equal to the heat released by its surroundings).
Enthalpy (H) - the heat content of a system at constant pressure
Thermochemical equation - a chemical equation that includes the enthalpy change (in a chemical equation, the enthalpy change for the reaction can be written as either a reactant or a product)
Heat of reaction - the enthalpy change for the chemical equation exactly as it is written
Heat of combustion - the heat of reaction for the complete burning of one mole of a substance
Molar heat of fusion - the heat absorbed by one mole of a solid substance as it melts to a liquid at a constant temperature
Molar heat of solidification - the heat lost when one mole of a liquid solidifies at a constant temperature
Molar heat of vaporization - the amount of heat necessary to vaporize one mole of a given liquid
Molar heat of condensation - the amount of heat released when one mole of vapor condenses at the normal boiling point (the quantity of heat absorbed by a vaporizing liquid is exactly the same as the quantity of heat released when the vapor condenses)
Molar heat of solution - the enthalpy change caused by dissolution of one mole of a substance (During the formation of a solution, heat is either released or absorbed)
Calorimeter - an insulated device used to measure the absorption or release of heat in chemical or physical processes
Hess's law of heat summation - if you add two or more thermochemical equations to give a final equation, then you also can add the heats of reaction to give the final heat of reaction (Hess's law allows you to determine the heat of reaction indirectly)
Standard heat of formation - the change in enthalpy that accompanies the formation of one mole of a compound from its elements with all substances in their standard states at 25 degrees celsius (For a reaction that occurs at standard conditions, you can calculate the heat of the reaction by using standard heat of formation)
Change in enthalpy - The heat gained or lost by a system at constant pressure. in this textbook, heat and enthalpy change are the same
Joules - unit for energy. One joule of heat raises the temperature of 1 g of pure water 0.2390 degree C
