Acid Disassociation Constant(Ka) - The Ratio of the concentration of the dissociated form of an acid to the undissociated form; stronger acids have larger Ka values than the weaker acids.
Acidic Solution - Any solution in which the hydrogen ion concentration is greater than the hydroxide ion concentration.
Alkaline Solution - A basic solution
Amphoteric - A substance that can act as both an acid and a base.
Base Dissociation Constant(Kb) - The ratio of the concentration of the conjugate acid times the concentration of the hydroxide ion to the concentration of the conjugate base.
Base Solution - Any solution in which the hydroxide ion concentration is greater than the hydrogen ion concentration
Buffers - A solution in which the hydroxide ion concentration is greater than the hydrogen ion concentration
Buffer Capacity - A measure of the amount of acid or base that may be added to a buffer solution before a significant change in pH occurs.
Conjugate Acid - The particle formed when a base gains a hydrogen ion
Conjugate Acid-Base Pair - Two substances that are related by the loss or gain of a single hydrogen ion
Diprotic Acid - Any acid that contains two ionizable protons
End Point - THe point in a titration at which the inidcator changes color
Equivalence Point - The point in a titration where the number of moles of hydrogen ions equals the number of moles of hydroxide ions
Hydronium Ion(H3O+) - The positive ion formed when a water molecule gains a hydrogen ion.
Ion-Product Constant for Water(Kw) - The product of the concentrations of hydrogen ions and hydroxide ions in water; It is 1 X 10^-14 at 25 degrees C
Lewis Acid - Any Substance that can accept a pair of electrons to forma covalent bond.
Lewis Base - Any substance that can donate a pair of electrons to form a covalent bond.
Monoprotic Acids - Any Acid that contains one ionizable proton.
Neutral Solution - An aqueous solution in which the concentration of hydrogen and hydroxide ions are equal; It has a pH of 7.0
Neutralization Reaction - A reaction in which an acid and a base react in an aqueous solution to produce a salt and water
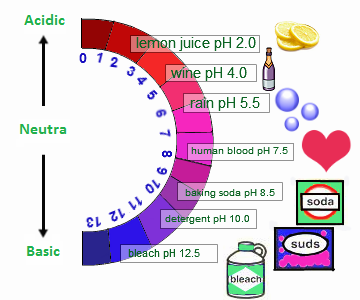
pH - A number used to denote the hydrogen-ion concentration, or acidity, of a solution; it is the negative logarithm of the hydrogen-ion concentration of a solution.
Salt hydrolysis - A process in which the cations of anions of a dissociated salt accept hydrogen ions from water or donate hydrogen ions to water
Self-Ionization - A term describing the reaction in which two water molecules react to produce ions
Standard Solution - A solution of known concentration used in carrying out a titration
Strong Acid - An acid that is completely (or almost completely) ionized in aqueous solution.
Strong Base - A base that completely dissociates into metal ions and hydroxide ions in aqueous solution.
Titration - Process used to determine the concentration of a solution (often an acid or base) in which a solution of known concentration (the standard) is added to a measured amount of the solution of unknown concentration until an indicator signals the end point.
Triprotic Acid - Any acid that contains three ionizable protons
Weak Acid - An acid that is only slightly ionized in aqueous solution
Weak base - A base that reacts with water to formth e hydroxide ion and the conjugate acid of the base
Acidic Solution - Any solution in which the hydrogen ion concentration is greater than the hydroxide ion concentration.
Alkaline Solution - A basic solution
Amphoteric - A substance that can act as both an acid and a base.
Base Dissociation Constant(Kb) - The ratio of the concentration of the conjugate acid times the concentration of the hydroxide ion to the concentration of the conjugate base.
Base Solution - Any solution in which the hydroxide ion concentration is greater than the hydrogen ion concentration
Buffers - A solution in which the hydroxide ion concentration is greater than the hydrogen ion concentration
Buffer Capacity - A measure of the amount of acid or base that may be added to a buffer solution before a significant change in pH occurs.
Conjugate Acid - The particle formed when a base gains a hydrogen ion
Conjugate Acid-Base Pair - Two substances that are related by the loss or gain of a single hydrogen ion
Diprotic Acid - Any acid that contains two ionizable protons
End Point - THe point in a titration at which the inidcator changes color
Equivalence Point - The point in a titration where the number of moles of hydrogen ions equals the number of moles of hydroxide ions
Hydronium Ion(H3O+) - The positive ion formed when a water molecule gains a hydrogen ion.
Ion-Product Constant for Water(Kw) - The product of the concentrations of hydrogen ions and hydroxide ions in water; It is 1 X 10^-14 at 25 degrees C
Lewis Acid - Any Substance that can accept a pair of electrons to forma covalent bond.
Lewis Base - Any substance that can donate a pair of electrons to form a covalent bond.
Monoprotic Acids - Any Acid that contains one ionizable proton.
Neutral Solution - An aqueous solution in which the concentration of hydrogen and hydroxide ions are equal; It has a pH of 7.0
Neutralization Reaction - A reaction in which an acid and a base react in an aqueous solution to produce a salt and water
pH - A number used to denote the hydrogen-ion concentration, or acidity, of a solution; it is the negative logarithm of the hydrogen-ion concentration of a solution.
Salt hydrolysis - A process in which the cations of anions of a dissociated salt accept hydrogen ions from water or donate hydrogen ions to water
Self-Ionization - A term describing the reaction in which two water molecules react to produce ions
Standard Solution - A solution of known concentration used in carrying out a titration
Strong Acid - An acid that is completely (or almost completely) ionized in aqueous solution.
Strong Base - A base that completely dissociates into metal ions and hydroxide ions in aqueous solution.
Titration - Process used to determine the concentration of a solution (often an acid or base) in which a solution of known concentration (the standard) is added to a measured amount of the solution of unknown concentration until an indicator signals the end point.
Triprotic Acid - Any acid that contains three ionizable protons
Weak Acid - An acid that is only slightly ionized in aqueous solution
Weak base - A base that reacts with water to formth e hydroxide ion and the conjugate acid of the base