Cracking - process by which hydrocarbons are broken down into smaller molecules
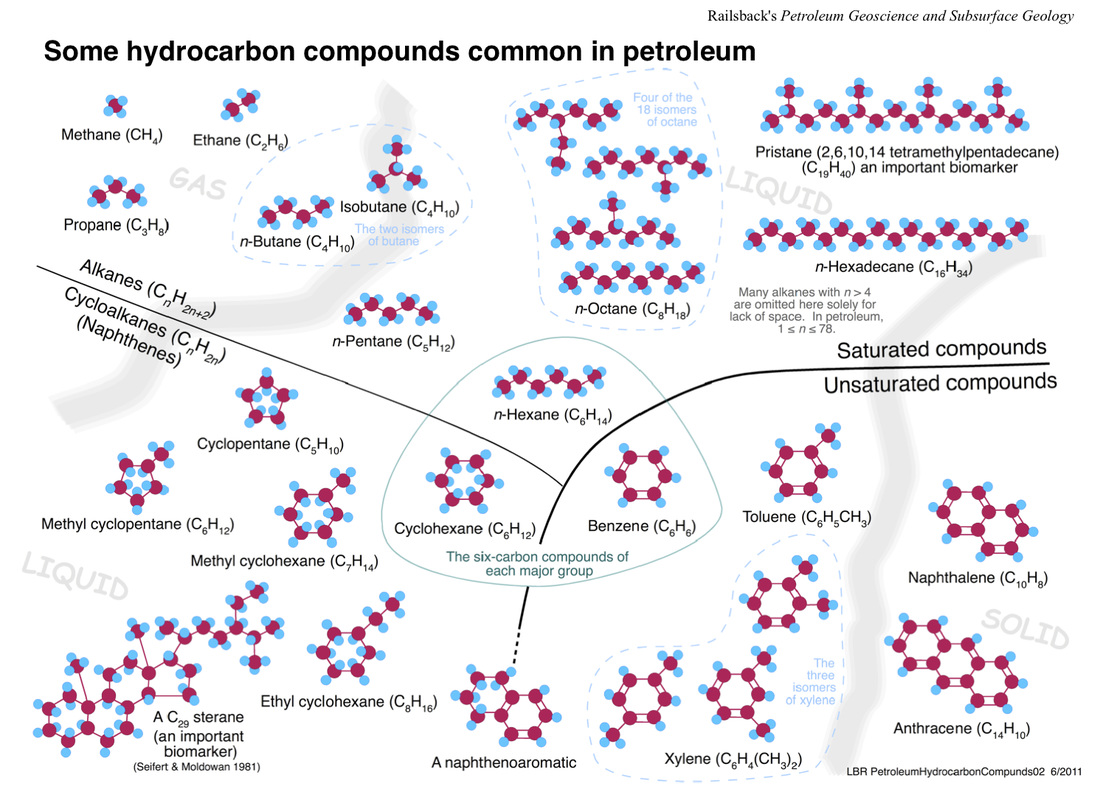
Petroleum - fossil fuel containing straight and branched chain alkanes
Distillation - process by which petroleum is separated into fractions
Lignite - Brown coal, consisting of about 50% carbon
Anthracite - Hard coal, which is high in carbon content
Alkyneshydro - carbons containing at least one carbon-carbon triple bonds. Other bonds may be single or double carbon-carbon bonds and single carbon-hydrogen bonds.
Substituent - Group of atoms that takes the place of hydrogen in a parent hydrocarbon
cis configuration - Geometric isomer with substituted groups on the same side of the double bond
homologous series - series of compounds related by a constant increment of change
arene - another name for an aromatic hydrocarbon
stereoisomers - molecules that in which the atoms are joined in the same order, but differ in the arrangement of atoms in space. Tow types of stereoisomers are geometric isomers and optical isomers.
saturated compounds - organic compounds that contain the maiximum number of hydrogen atoms per carbon atom
cyclic hydrocarbons - organic compounds that contain hydrocarbon rings
resonance - when two or more equally valid structures can be drawn for a molecule
phenyl group - name given to a benzene ring when it is a substituent
aromatic compounds - group of hydrocarbons that contain a benzene ring or a ring with bonding like that of benzene
isomers - compounds that have the same molecular formula but different molecular structures
structural isomers - compounds that have the same molecular formula but the atoms are joined in a different order. Structural isomers differ in physical properties such as boiling point and melting point. They also have different chemical reactivities.
asymmetric carbon - a carbon with four different groups attached
geometric isomers - isomers that differ in the concentration of groups around a double bond
trans configuration - configuration of the substituted groups on opposite sides of the double bond
optical isomers - pairs of molecules that differ only in the way four different groups are arranged around a central carbon atom
alkenes - are hydrocarbons that contain at least one carbon-carbon double bond. Other bonds may be single carbon-carbon bonds and carbon-hydrogen bonds (a carbon-carbon double bond is shown as a parallel line in a structural formula)
unsaturated compounds - organic compounds that contain double or triple carbon-carbon bonds
straight chain alkanes - organic compounds that contain only carbon and hydrogen
branched chain alkanes - alkanes that contain one or more alkyl substituents
alkyl group - a hydrocarbon substituent
hydrocarbons - hydrocarbons that contain only single covalent bonds
Aliphatic Hydrocarbons - any straight-chain or branched-chain alkanes, alkenes, and alkynes
alkanes - A hydrocarbon in which there are only single covalent bonds. In any alkane, all the carbon-carbon bonds are single covalent bonds, and all the other bonds are carbon-hydrogen bonds. The carbon atoms in an alkane can be arranged in a straight chain or in a chain that has branches.
condensed structural formulas - a structural formula that leaves out some bonds and or atoms, the presence of these atoms or bonds is understood
carbonbecause carbon has four valence electrons, a carbon atom always forms four covalent bonds
properties of alkanes - molecules of hydrocarbons, such as alkanes, are nonpolar molecules. The attractions between nonpolar molecules are weak van der Waals forces. So alkanes of low molar mass tend to be gases or liquids that boil at a low temp.
benzene - in a benzene molecule, the bonding electrons between carbon atoms are shared evenly around the ring.
natural gas - an important source of alkanes of low molar mass.
petroleum - the refining process starts with the distillation of petroleum (crude oil) into fractions according to boiling point.
coal - is formed when tree ferns and mosses die. its classified by its hardness and carbon content. It consists largely of condensed aromatic compounds of extremely high molar mass. These compounds have a high proportion of carbon compared with hydrogen.
Petroleum - fossil fuel containing straight and branched chain alkanes
Distillation - process by which petroleum is separated into fractions
Lignite - Brown coal, consisting of about 50% carbon
Anthracite - Hard coal, which is high in carbon content
Alkyneshydro - carbons containing at least one carbon-carbon triple bonds. Other bonds may be single or double carbon-carbon bonds and single carbon-hydrogen bonds.
Substituent - Group of atoms that takes the place of hydrogen in a parent hydrocarbon
cis configuration - Geometric isomer with substituted groups on the same side of the double bond
homologous series - series of compounds related by a constant increment of change
arene - another name for an aromatic hydrocarbon
stereoisomers - molecules that in which the atoms are joined in the same order, but differ in the arrangement of atoms in space. Tow types of stereoisomers are geometric isomers and optical isomers.
saturated compounds - organic compounds that contain the maiximum number of hydrogen atoms per carbon atom
cyclic hydrocarbons - organic compounds that contain hydrocarbon rings
resonance - when two or more equally valid structures can be drawn for a molecule
phenyl group - name given to a benzene ring when it is a substituent
aromatic compounds - group of hydrocarbons that contain a benzene ring or a ring with bonding like that of benzene
isomers - compounds that have the same molecular formula but different molecular structures
structural isomers - compounds that have the same molecular formula but the atoms are joined in a different order. Structural isomers differ in physical properties such as boiling point and melting point. They also have different chemical reactivities.
asymmetric carbon - a carbon with four different groups attached
geometric isomers - isomers that differ in the concentration of groups around a double bond
trans configuration - configuration of the substituted groups on opposite sides of the double bond
optical isomers - pairs of molecules that differ only in the way four different groups are arranged around a central carbon atom
alkenes - are hydrocarbons that contain at least one carbon-carbon double bond. Other bonds may be single carbon-carbon bonds and carbon-hydrogen bonds (a carbon-carbon double bond is shown as a parallel line in a structural formula)
unsaturated compounds - organic compounds that contain double or triple carbon-carbon bonds
straight chain alkanes - organic compounds that contain only carbon and hydrogen
branched chain alkanes - alkanes that contain one or more alkyl substituents
alkyl group - a hydrocarbon substituent
hydrocarbons - hydrocarbons that contain only single covalent bonds
Aliphatic Hydrocarbons - any straight-chain or branched-chain alkanes, alkenes, and alkynes
alkanes - A hydrocarbon in which there are only single covalent bonds. In any alkane, all the carbon-carbon bonds are single covalent bonds, and all the other bonds are carbon-hydrogen bonds. The carbon atoms in an alkane can be arranged in a straight chain or in a chain that has branches.
condensed structural formulas - a structural formula that leaves out some bonds and or atoms, the presence of these atoms or bonds is understood
carbonbecause carbon has four valence electrons, a carbon atom always forms four covalent bonds
properties of alkanes - molecules of hydrocarbons, such as alkanes, are nonpolar molecules. The attractions between nonpolar molecules are weak van der Waals forces. So alkanes of low molar mass tend to be gases or liquids that boil at a low temp.
benzene - in a benzene molecule, the bonding electrons between carbon atoms are shared evenly around the ring.
natural gas - an important source of alkanes of low molar mass.
petroleum - the refining process starts with the distillation of petroleum (crude oil) into fractions according to boiling point.
coal - is formed when tree ferns and mosses die. its classified by its hardness and carbon content. It consists largely of condensed aromatic compounds of extremely high molar mass. These compounds have a high proportion of carbon compared with hydrogen.
